 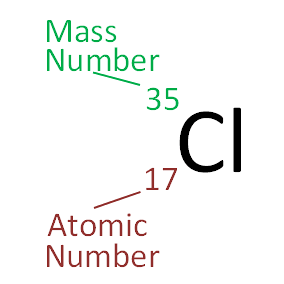
To find the atomic mass of chlorine, the atomic mass of each isotope is multiplied by the relative abundance (the percent abundance in decimal form) and then the individual masses are added together. The atomic mass is found by multiplying the mass of the isotope by its relative abundance, then adding the individual masses together.ġ.Ĝalculate the atomic mass of chlorine using the information provided in the following table. Isotopes occurring in higher percentages will have a larger effect on the atomic mass, while isotopes occurring in lower percentages will have a smaller effect. The isotopes of an element do not occur in equal percentages in nature, so a weighted average must be taken in order to achieve the atomic mass of the element. Atomic mass is measured in atomic mass units (amu), where one amu is roughly equivalent to the mass of a single proton or neutron. The number of neutrons is not equal so the masses of the isotopes will not be the same. 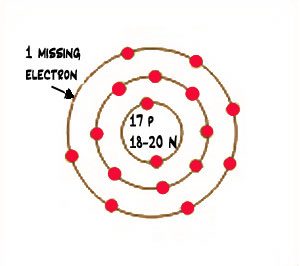
The number of protons determines the identity of the atom, and isotopes have identical atomic numbers, so the atoms are of the same element. Isotopes are atoms that have the same atomic number (# of protons) but a different number of neutrons. The atomic mass of an element is equal to the weighted average of the isotopes for that element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
